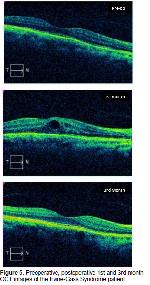
This study revealed that the mean CMT significantly increased 1 week, 1, and 3 months after uneventful phacoemulsification surgery even in patients with no risk factors for IGS. The increase reached its peak level in the postoperative 1st month and showed a declining trend in the postoperative 3rd month. We detected IGS in one of the patients (%2) of this study in postoperative 1st month and the PCME was dissolved with topical NSAID treatment in the 3rd-month examination after surgery.
The release of inflammatory cytokines such as prostaglandin and arachidonic acid leads to the breakdown of the blood-retinal barrier which causes the CME in IGS 20,21. Besides, a positive correlation between postoperative anterior camera flare and macular thickness was previously reported which points out the inflammatory mechanisms behind IGS 22. However, various hypotheses were proposed to explain the triggers of inflammation. Firstly, the surgical trauma, particularly iris contact, was thought to be associated with the increased inflammation 20,23. Taipale et al reported that utilization of pupil expansion devices in cataract surgeries were related to increased risk for PCME 24. Moreover, these devices require longer surgery times which may also alleviate the inflammation. However, another study revealed that Femto-assisted cataract surgeries in which the anterior chamber manipulations are minimal, CMT alterations were also significantly reduced 25. In our study, although the surgery times of experienced surgeons were under 15 minutes and no pupil expansion device was applied, we still observed significantly increased CMT. Therefore, it is conceivable that surgical trauma may not be the only factor, even if it affects the development of the IGS. The microscope light source, which is an indispensable equipment of all surgeries, were angiographically demonstrated to cause remodeling of parafoveal vessels 26. Considering the parafoveal location of CME in the IGS, the retinal photochemical alterations induced by the microscope illumination may explain the vascular leakage.
The PCME after uneventful cataract surgery was previously demonstrated even in patients with no predisposing factors 6. Although surgical maneuvers and light exposure occur in all surgeries, only very few patients suffer IGS. In an animal model, Xu et al reported that surgical interventions induce expression of chemokine ligand 2 and increased interleukin-1β upregulation in choroid and retina 27. Therefore, individual differences of gene expressions in retinal layers may explain the tendency to face this complication.
Several studies reported that the CMT peak occurs 4 to 6 weeks after cataract surgery 28,29. Consistently, we also detected the highest CMT values in the postoperative 1st-month measurements. However, despite the increase in CMT after surgery, CDVA levels were also significantly increased over time. The mean CMT elevation is usually subclinical and does not affect visual acuity 30. It was reported that the increase in CMT after cataract surgery reaches preoperative values spontaneously in an average of 2 to 6 months 9,28. In our study, the CMT values tended to decrease in the 3rd month after surgery, however that was still significantly higher compared to preoperative values. Besides, we observed that the CMT values were comparable in the 1st week and 3rd month after surgery. The cataract formation blocks 18-40% of the light reaching the retina and that may cause different OCT measurements due to its light scattering effect 30,31. Hence, IOL and the new optic media after surgery may also alter the measurements.32. In our study, we also observed significant decreases in IOP in the 1st week and 1st month. The retinal vasculature alterations caused by the decreased IOP may also lead to a parafoveal leak 33,34. However, Zhou et al did not find a correlation between the IOP and CMT 26. Similarly we also did not detect significant correlation between these two parameters in any of the examinations. Therefore, further studies are needed to explain the relationship between IOP change and CME development.
The prevalence of IGS after phacoemulsification surgery was reported between 0.1-2.3% in various studies 21,35,36. Although there was no risk factor, a 66-year-old male patient (2%) in this study suffered IGS in the 1st month after uneventful phacoemulsification surgery. This rate is consistent with the current literature. The PCME disappeared by topical NSAID treatment and the CMT was decreased and the CDVA was improved in the 3rd-month examination. Considering the role of inflammation in the development of IGS, topical corticosteroid eye drops alone may sometimes fail to control the postoperative inflammation. Zaczek et al. observed lower CMT values in patients receiving topical NSAID treatment after phacoemulsification surgery 37. Similarly, Tzelikis et al. reported that topical NSAIDs significantly reduced CMT but did not make a significant impact on CDVA 38. Although postoperative topical NSAID treatment was recommended in patients with known risk factors such as uveitis, VMT, diabetes, or ERM, it is controversial to recommend these medications in all cases as a routine postoperative regimen due to their ocular surface side effects 39. Besides, it was reported that postoperative CMT increase may spontaneously regress in 80-95% of cases within 3-12 months 6,40. Even though there are plenty of topical NSAID options, topical Nepafenac was shown to be safe and effective in several studies to manage the post-phacoemulsification inflammation 24,38. Nevertheless, as in our case, topical Nepafenac treatment may accelerate the regression of CME in IGS cases.
This study had several limitations. Firstly, the CMT might have been detected higher or the IGS lower since the patients were followed for only 3 months and the normalization of CME may proceed up to 12 months. Secondly, none of the patients in our study routinely received topical NSAID medications, so that, it was not possible to fully evaluate the effect of this treatment on CMT. Although we obtained favorable outcomes with topical nepafenac treatment in the IGS patient, it is recommended to evaluate the efficacy of different molecules in IGS with randomized controlled studies. Finally, due to the small sample size, it may not be suitable to generalize our results. Further studies in different postoperative treatment regimens in larger groups may provide a better understanding of the mechanisms behind IGS.
In conclusion, we observed that the CMT significantly increased in the 1st week, 1st month, and 3rd month after surgery compared to the preoperative values in patients with no risk factors who underwent uneventful phacoemulsification surgery. The difference was significant compared to the fellow eye particularly in the 1st month which revealed the highest mean postoperative CMT. Besides, we observed that IGS, which occurred in a patient who did not receive postoperative topical NSAID treatment, disappeared in 2 months with topical nepafenac medication. However, more comprehensive studies are needed to recommend a routine postoperative topical NSAID regimen after phacoemulsification surgery.