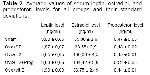A total of 38 female Wistar Albino rats having average weight of 200 g (180-220) were used in the study. Rats were preserved in the laboratory of test animals of our faculty. Rats were hold in plastic cages in four and five groups in the laboratory with air conditioning system. Ambient kept in room temperature. Rats hold in 12 hours light and 12 hours darkness. Rats were fed with ready pellets in special steel pots and their waters were given by ball feeding bottle made from stainless steel. The cages of rats were cleaned daily. Rats were divided into 5 groups randomly:
Group 1 (n = 7): In this group, sham (only abdomen opened and closed, ovariectomy was not made) operation was applied to rats with 206 gr. average weight under general anesthesia (giving Rompun/Ketamine; ¾, 0.4 ml/rat im). Rats received no medical treatment. At the end of the study, decapacitation was made and their serums were taken in glass tubes.
Ovariectomy
Ovariectomy was performed in 31 rats other than rats in sham group. After application of general anesthesia (giving Rompun/Ketamine; ¾, 0,4 ml/rat im), the hairs on lower abdomen region of rats were shaved, abdomen was opened through a midline incision. Tubes were found and connected bilaterally and ovaries were taken out. After completing operation, the abdomen of rats were closed and waited for their healing. Rats in these groups were divided into four subgroups following the healing period.
Group 2 (n = 9): In this group, ovariectomy was performed in the way that was mentioned previously to rats with an average weight of 246 gr. After 3 months of healing period, 0,2 ml/rat/day SF was applied under skin during one month. Decapacitation was made at the end of one month and blood was taken to normal glass tubes.
Group 3 (n = 7): In this group, ovariectomy with the same technique was performed to rats having an average weight of 260 gr. and after 3 months healing period, 17β Estradiol in the dose of 30 mg/kg/day was administered under skin for a period of one month. Decapacitation was made at the end of one month and blood sample was taken in normal glass tubes from each rat.
Group 4 (n = 7): In this group, rats had an average weight of 262 gr. After 3 months of a healing period following ovariectomies, 30 mg/kg/day 17β Estradiol + 2 mg/rat/day progesterone was given under skin for one month. Blood sample was obtained by decapitation at the end of one month.
Group 5 (n = 8): Rats in this group with an average weight of 240 gr underwent ovariectomy and after 3 months of healing period, 12,5 mg/day intraperitoneal vitamin E was administered for one month. Decapacitation was made at the end of one month and blood was taken in normal glass tubes.
Rats were weighed before operation, before giving the test agents and before predecapacitation. In order to prevent the formation of stress, decapitation was performed within 5-10 seconds. While consequtive decapitation operations were made, guillotine was washed by tap water, cleaned and dried before each operation. The bloods of animals taken by decapitation were placed in normal glass tubes, and kept at +4 ºC in 10 minutes. It was centrifuged for 10 minutes. Serums separated were preserved at –20 ºC in deep freeze to measure leptin, estradiol and progesterone levels.
The measurement of leptin, estradiol and progesteron levels in blood serum samples taken from rats was made by radioimmune assay system.
In statistical examination, at first it has been assessed that whether data shows normal in-group distribution by Sample Kolmogorov Smirnov Z test from nonparametric tests. When it was shown that all groups have normal distribution, the comparison among groups were made by One Way ANOVA test. Data was analyzed by post-hoc Tukey test. Significance level was accepted p < 0.05. The analysis of data was made by SPSS 15 Windows packaged software.